How personalized medicine is transforming your health care
PHOTOGRAPH BY CRAIG CUTLER (HAND WITH CHIP); SAMUEL SANCES, CEDARS-SINAI (BACKGROUND)
READ CAPTION
PRECISION-MEDICINE-MICRO-CHIP-STEM-CELLS
On this chip, researchers Clive Svendsen and Samuel Sances at Cedars-Sinai in Los Angeles… Read More
PHOTOGRAPH BY CRAIG CUTLER (HAND WITH CHIP); SAMUEL SANCES, CEDARS-SINAI (BACKGROUND)
Stunning advances in gene research and data mining will predict diseases and devise treatments tailored to each of us.
16 MINUTE READ
BY FRAN SMITH
PHOTOGRAPHS BY CRAIG CUTLER

This story appears in the January 2019 issue of National Geographic magazine.
TWELVE YEARS AFTER Teresa McKeown beat stage 3 breast cancer with a brutal regimen of chemotherapy and a double mastectomy, the disease returned, more aggressively than before. This time chemotherapy failed. Day after day, she sat in a chair in her living room, too sick to move. She kept four journals, one each for her husband and her three grown children, and mustered the strength to write her thoughts about a future she didn’t expect to share.
She withered to 98 pounds because tumors in her bowel made it almost impossible to eat. McKeown is not one to rage or panic, but before surgery to remove the blockage, she made a rare admission of anguish. “I am so praying that if things are not going to end well for me or there is a complication following this surgery, that I just pretty quickly pass away,” she recalled telling her older daughter. “I don’t know how much more pain I can tolerate.”
this background color div is for when the page loads
end bg color the background image itself should be added to only this div
end bg image
STATIC FIELDS
Get more of the inspiring photos and stories we’re known for, plus special offers.
CUSTOMER INPUT FIELDS
SUBMIT BUTTON
By signing up for this email, you are agreeing to receive news, offers, and information from National Geographic Partners, LLC and our partners. Click here to visit our Privacy Policy. Easy unsubscribe links are provided in every email.
Desperate and determined, she asked her surgeon, Jason Sicklick, if he knew of any experimental treatments that might buy her more time. As it happened, he is a co-leader of a study at the cutting edge of what’s come to be called precision, or personalized, medicine.
The approach, built on advances in gene research and data analytics, holds transformative possibilities for cancer treatment and could upend the way medicine traditionally has been practiced. Rather than lump patients together under broad categories of diseases, precision medicine aims to tailor prevention, diagnosis, and treatment to a person’s unique biochemical makeup.
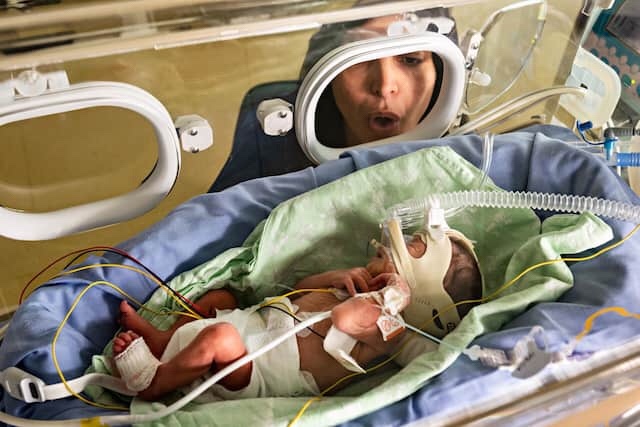
A MOTHER’S VOICE
Cristina Iossa sings to her prematurely born son, Alessandro, at the neonatal intensive care unit of University Hospital in Modena, Italy. In NICUs, parents are now a more frequent presence. “The mother’s voice to her offspring is one of the most primal precision medicines of all, because it primarily calls you and nobody else,” says Manuela Filippa, a researcher at the Universities of Valle d’Aosta and Geneva. Scientists theorize that exposure to a mother’s voice stimulates a newborn’s brain to develop optimally to interpret sounds and understand language.
McKeown joined I-PREDICT, a precision cancer study at the University of California, San Diego-affiliated Moores Cancer Center. Researchers there don’t rely on any particular therapy. Instead they analyze the DNA in a patient’s cancer cells. Using special algorithms, a computer then scours data on thousands of gene variants, hundreds of anticancer drugs, and millions of drug combinations to find the treatment that best targets the tumor’s abnormalities. It may be a new immunotherapy, old-line chemotherapy, hormonal therapies, or drugs that aren’t specifically approved for cancer.
“It’s a very simple principle,” said Razelle Kurzrock, an oncologist and director of the Moores Center for Personalized Cancer Therapy. “You pick the right drugs for each patient based on the tumor profile, not based on a part of the body or based on what type of cancer 100 other people have. It’s all about that patient sitting in front of me.”
McKeown’s tumors were riddled with different mutations. “These are the kinds of patients we used to hang our heads and feel sorry for,” Kurzrock said. But they’re among the best candidates for a new class of immunotherapies called checkpoint inhibitors. The drugs prevent tumor-made proteins from binding to immune cells and shutting them down, which restores the patient’s ability to fight the cancer. More mutations mean the reactivated immune cells have more targets to attack and eradicate.
I-PREDICT matched McKeown with nivolumab, a checkpoint inhibitor approved for advanced melanoma, kidney cancer, and certain lung cancers but not for breast cancer. After two infusions, the tumor markers in her blood declined by more than 75 percent. Four months later, after additional infusions, tests detected no evidence of cancer.

SPECIAL ISSUE
THE FUTURE OF MEDICINE
She gave her body to science. Her corpse became immortal.
As biomedicine advances, are we clear on the implications?
Spending more on health care may not lengthen life the most
On a hot summer day, a year and a half after she entered the trial, McKeown, 57, showed me around her garden in Valley Center, California. It’s a three-acre expanse of lawns, blooming trees, and rosebushes of red, white, lavender, orange, and brilliant yellow nestled, improbably, amid the parched, scrub-covered hills.
“I just feel so grateful,” she said. “I love this whole notion of individualized medicine. I love that they’re figuring out what’s causing that mutation and how to target it, as opposed to chemo that’s so disruptive across the board. Can we just get there faster?”
Precision medicine flips the script on conventional medicine, which typically offers blanket recommendations and prescribes treatments designed to help more people than they harm but that might not work for you. The approach recognizes that we each possess distinct molecular characteristics, and they have an outsize impact on our health.

Around the world, researchers are creating precision tools unimaginable just a decade ago: superfast DNA sequencing, tissue engineering, cellular reprogramming, gene editing, and more. The science and technology soon will make it feasible to predict your risk of cancer, heart disease, and countless other ailments years before you get sick. The work also offers prospects—tantalizing or unnerving, depending on your point of view—for altering genes in embryos and eliminating inherited diseases.
More immediately, the research points the way to customized therapies for the most recalcitrant cancers. Last spring, researchers at the National Cancer Institute reported the dramatic recovery of a woman with metastatic breast cancer, Judy Perkins, after an experimental therapy using her own immune cells to attack her tumors. The team, led by Steven Rosenberg, an immunotherapy pioneer, had sequenced her tumor’s DNA to analyze the mutations. The team also extracted a sampling of immune cells called tumor-infiltrating lymphocytes and tested them to see which ones recognized her tumor’s genetic defects. The scientists reproduced the winning lymphocytes by the billions and infused them into Perkins, along with a checkpoint inhibitor, pembrolizumab. More than two years later, Perkins, a retired engineer from Florida, shows no signs of cancer.


CANCER KILLERS
Judy Perkins is surrounded by white blood cells known as tumor-infiltrating lymphocytes, or TILs, which cured her breast cancer (see CT scans below). When Perkins was first diagnosed, she had her left breast removed, but the cancer returned. Despite chemotherapy, hormonal treatments, and targeted therapies, a new tumor grew in her chest. When the cancer spread, she was given months to live. But in an experimental treatment devised by Steven Rosenberg at the National Cancer Institute, Perkins was infused with 82 billion of her own TILs, which she calls her “army.”
start interactive embed
YOUR GRAPHIC CODE GOES HERE SAMPLE STYLES. REMOVE ! TO SEE THIS Remove the following line if NOT using ai2html Generated by ai2html v0.78.0 – 2018-12-04 09:43 ai file: ngm-1901-PrecisionMedicine-CrossSection_ai2html.ai
Artboard: mobile
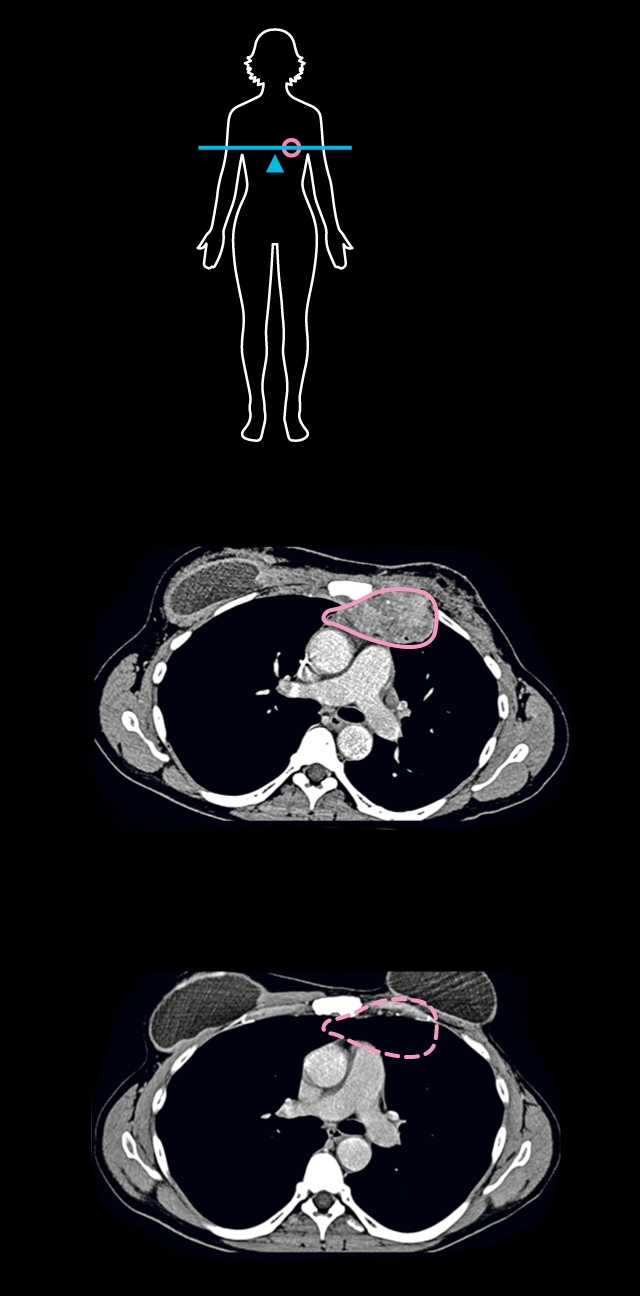
Cross sections
(seen from
below)
show the
treatment
eliminated
the tumor.
BEFORE TREATMENT
Tumor
28 MONTHS AFTER
End ai2html – 2018-12-04 09:43 END GRAPHIC CODE
end interactive embed
CT SCANS: STEVEN ROSENBERG, NATIONAL CANCER INSTITUTE
One success story, of course, doesn’t make a medical revolution. Two other patients in Rosenberg’s trial died. “I’m a little point of light,” Perkins said. “We need a lot more points of light to get the data to be able to harness the immune system.”

The game changer may not be this treatment but what it says about the power of precision medicine. The distinctive mutations that fuel a person’s cancer may be its undoing.
Thirty years ago, scientists thought that it would be impossible to crack our genetic code and sequence the 3.2 billion pairs of compounds in our DNA. “It was like you were talking fairy tales,” Kurzrock said. “The conventional wisdom was that it would never happen. Never! And then in 2003, never was over.”
It took the Human Genome Project 13 years, roughly one billion dollars, and scientists from six countries to sequence one genome. Today sequencing costs about a thousand dollars. The latest machines can churn out the results in a day. The technology, combined with sophisticated molecular analysis, illuminates the astonishing biochemical variations that make every human body unique.
start interactive embed
YOUR GRAPHIC CODE GOES HERE SAMPLE STYLES. REMOVE ! TO SEE THIS Remove the following line if NOT using ai2html Generated by ai2html v0.78.0 – 2018-12-05 16:58 ai file: ngm-1901-precision-medicine-immunity_ai2html.ai
Artboard: NGM-Spread Artboard: Artboard_2

SUPERCHARGED
IMMUNITY
Researchers are pioneering cancer treatments that go beyond creating new drugs. The National Cancer Institute is testing a form of immunotherapy that identifies mutations in a patient’s tumors and then unleashes the immune system to attack them. Here’s how the technique worked for Judy Perkins, whose cancer was eliminated.
Perkins’s tumor was
genomically sequenced,
identifying 62 mutations.
TTTTAAGATCCAATGATCTTCAAA
ACGCTGCAAGATTCTCAACCTGC
TTTACTAAGCGCTGGGTCCTACTC
CAGCGGGATTTTTTATCTAAAGA
CGATGAGAGGAGTATTCGTCAGA
CCACATAGCTTTCATGTCCTGATC
GGAAGGATCGTTGGCGCCCGACC
CTCAGACTCTGTAGTGAGTTCTAT
GTCCGAGCCATTGCATGCGAGAT
CGGTAGATTGATAGGGGATACAG
AATATCCCTGGATGCAATAGACG
GACAGCTTGGTATCCTAAGCGTA
GTCGCGCGTCCGAACCCAGCTCT
ACTTTAGAGGCCTCGGATTCTGG
TGCCCGCAGGCCGCAGAACCGAT
TAGGGGCATGTACAACAATATTT
ATTAGTCACCTTTGAGACACGAT
CTCCCACCTCACTGGAATTTAGTT
CCTGCTATAATTAGCCTTCCTCAT
Doctors found immune
cells called lymphocytes
that attacked four mutations.
TTTTAAGATCCAATGATCTTCAAA
ACGCTGCAAGATTCTCAACCTGC
TTTACTAAGCGCTGGGTCCTACT
CCAGCGGGATTTTTTATCTAAAG
ACGATGAGAGGAGTATTCGTCAG
ACCACATAGCTTTCATGTCCTGAT
CGGAAGGATCGTTGGCGCCCGAC
CCTCAGACTCTGTAGTGAGTTCT
ATGTCCGAGCCATTGCATGCGAG
ATCGGTAGATTGATAGGGGATAC
AGAATATCCCTGGATGCAATAGA
CGGACAGCTTGGTATCCTAAGCG
TAGTCGCGCGTCCGAACCCAGCT
CTACTTTAGAGGCCTCGGATTCT
GGTGCCCGCAGGCCGCAGAACCG
ATTAGGGGCATGTACAACAATAT
TTATTAGTCACCTTTGAGACACG
ATCTCCCACCTCACTGGAATTTAG
TTCCTGCTATAATTAGCCTTCCTC
The tumor-infiltrating
lymphocytes were
isolated and cultured.
These lymphocytes
were replicated, generating billions
of her own cancer-fighting cells.
Once infused into
Perkins’s body, the cells
destroyed the cancer.
JASON TREAT, NGM STAFF; KELSEY NOWAKOWSKI
ART: CHRISTOPHER DELORENZO. SOURCE: STEVEN
ROSENBERG, NATIONAL CANCER INSTITUTE
End ai2html – 2018-12-05 16:58 END GRAPHIC CODE
end interactive embed
The more scientists discover about those differences, the cruder conventional medicine seems. Consider one-pill-fits-all prescribing. Most people who take a blockbuster drug, such as a statin or corticosteroid, benefit. But genomics reveals that many people don’t. The Food and Drug Administration has identified about a hundred drugs that may not work as commonly prescribed in people with specific gene variants.
The problem can be deadly. The drug clopidogrel, for instance, is routinely given to prevent blood clots in patients after a heart attack. But about a quarter of the population has a gene variant that produces a defective form of an enzyme needed to activate the drug. Alan Shuldiner, a professor of medicine and a genetics researcher at the University of Maryland, found that when those people are prescribed the drug, they are twice as likely to have a repeat attack or die within a year of the first, compared with patients who don’t have the variant. Some major medical centers now screen heart attack patients for the variant, but the test is far from routine.
Many experts say that a decade from now, a DNA profile will be part of everyone’s medical record. Geisinger, a large health system in Pennsylvania and New Jersey, recently began offering genome sequencing as a routine part of preventive care, along with mammograms and colonoscopies.

Like advances in computer chips, which liberated us from desks and then tethered us to smartphones, the shift to genomics and data-driven medicine will be disruptive in unpredictable—and perhaps distressing—ways. We soon will have at our fingertips extensive data about diseases we may develop over the course of our lifetimes.
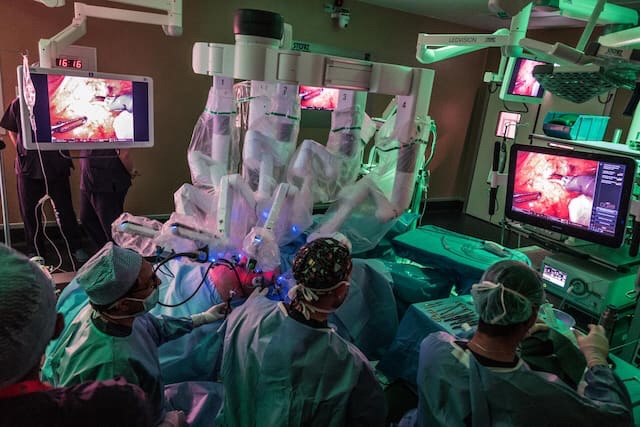
TUMOR-FIGHTING AVATARS
Surgeons remove a patient’s malignant tumor at the Champalimaud Foundation in Lisbon, Portugal, with a da Vinci Xi robot’s micro-instruments. Some of the cancer cells will be transplanted to zebrafish larvae and treated with the patient’s chemotherapy regimen to test its effectiveness (shown in the following photos).
To glimpse what that future might look like, meet geneticist Michael Snyder. He directs Stanford University’s Center for Genomics and Personalized Medicine, and for the past nine years he has been tracking molecular and physiological markers in his body. The result is a high-definition depiction of his inner workings that registers fluctuations that may signal problems. It’s like a weather map, charting shifts in the atmosphere to predict storms.

Snyder and the team in his lab take his DNA sequence into account as they analyze a continuous data stream. It includes measurements from blood, urine, and stool specimens he routinely provides and readings from bio-sensors he wears on both wrists, his ring finger, and his right arm. His team tracks his gene expression, proteins and metabolites, and physiological measures such as his exercise activity, heart rate, skin temperature, and blood oxygen. He undergoes MRIs, echocardiograms, and other scans to detect changes in his organs, muscles, and bone density.

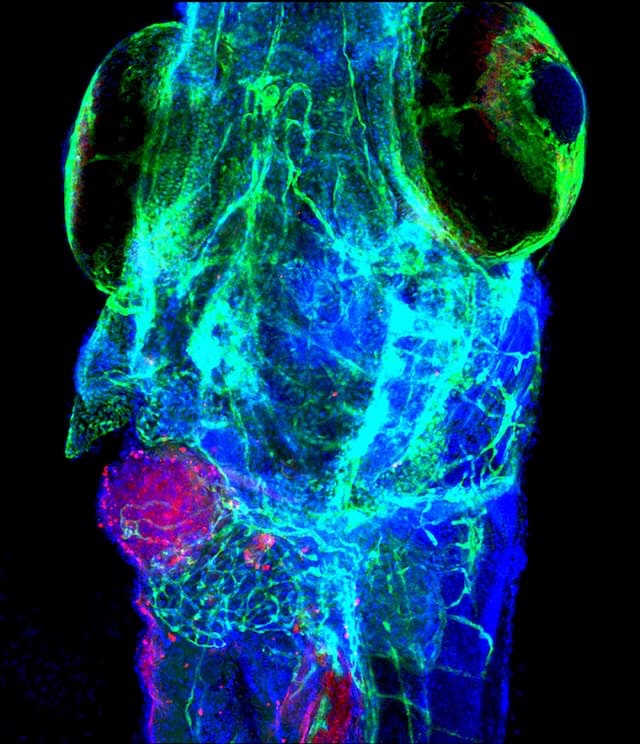
Top: Zebrafish could become a powerful tool for identifying the best chemotherapy to kill a particular patient’s cancer. Biologists Rita Fior and… Read More
PHOTOGRAPH BY RITA FIOR, CHAMPALIMAUD FOUNDATION (BOTTOM)

Snyder is not a hypochondriac. At 63, he’s lean, and if not for his receding hairline, he could be mistaken for a postdoc half his age. He has devised ways to perform genetic and molecular analyses, starting a dozen biotech companies. At Stanford, he’s trying to apply this work to detecting illness at its earliest stages, before it causes problems. He became his own guinea pig because he didn’t think anyone else would stick with all the tests and monitoring.
“Who knew I would turn out to be so interesting!” he said.
Four years ago, his sensors picked up an infection, through changes in his heart rate and blood oxygen level, before he felt sick. When he developed a fever, he suspected Lyme disease. By the time the standard test confirmed his hunch, he had already finished a course of antibiotics.
start interactive embed
YOUR GRAPHIC CODE GOES HERE SAMPLE STYLES. REMOVE ! TO SEE THIS Remove the following line if NOT using ai2html Generated by ai2html v0.66.4 – 2018-12-12 09:42 ai file: ngm-1901-precision-medicine-human-machine-graphic_ai2html.ai
Artboard: mobile

Continuously Monitoring the Human Machine
While precision medicine tailors treatment to one person, precision health uses detailed data to help people adapt their behavior and make healthy lifestyle choices. The goal: to avoid the need for treatment in the first place by passively collecting, transmitting, and storing health information as an individual goes about daily life.
While sleeping
Advances in electrodes small and flexible enough to fit in textiles could lead to pillow- cases and sheets able to monitor brain waves and sleep patterns.
Upon waking
Toilets that check urine and stool for disease are being developed. In the future, smart mirrors could measure vital signs with radar, and toothbrushes might analyze saliva.
In the kitchen
Smart refrigerators might soon monitor the food stored and record its nutritional informa- tion. Food quality and freshness would be tracked, along with dietary habits.
Plugged in
Smartphones could analyze patterns that might indicate depression—such as a drop-off in social communication—and alert the user to address potential mental health issues.
In the car
Sensors could warn a driver about dangerous pollution levels, high blood alcohol content detected on the breath, and driving that indicates stress and drowsiness.
On the run
Electronics like Fitbits or electric membranes attached to the skin can track exercise, vital signs, and ultraviolet exposure, while a “smart bra” might detect breast cancer.
In the lab
“Electronic noses” could detect volatile organic compounds in the breath or in secretions such as sweat and saliva to find “smellprints” of diseases, including lung and ovarian cancer.
In your body
Contact lenses may soon check pressure and
glucose levels in the eye. Implantable technologies already monitor cardiac activity, nitrogen, and oxygen levels.
A positive feedback loop
An individual’s health risk factors are assessed, then compared with large-scale population data, which can suggest beneficial choices or interventions. Passive monitoring throughout the day is key to constantly improving outcomes.
Risk
assess- ments
Data
portal
Monitoring
• Family history
• Genetic screening
• Socioeconomic factors
• Environmental factors
• Machine learning
• Pattern recognition
• Early medical intervention
• Behavioral changes
JASON TREAT, NGM STAFF; KELSEY NOWAKOWSKI
ART: CHRISTOPHER DELORENZO
SOURCE: SANJIV SAM GAMBHIR, STANFORD UNIVERSITY
Artboard: desktop-small
End ai2html – 2018-12-12 09:42 END GRAPHIC CODE
end interactive embed

He also watched himself develop type 2 diabetes. His DNA had shown a predisposition, but he’d dismissed it because he was slim and had no family history of the disease. After a nasty viral infection, his glucose level shot up and stayed high, so he thought he might have diabetes. His doctor initially brushed off the possibility, as he had, but tests confirmed the disease.
He gave up sweets, doubled his bicycling, and started running four miles four times a week. He tracked the foods that spiked his glucose—“barbecued pork,” he moaned sorrowfully—and adjusted accordingly. Over nine months his glucose returned to normal. Meanwhile he has recruited more than a hundred volunteers for similar profiling.
I signed up and went to Stanford for a battery of tests, including genome sequencing. Orit Dagan-Rosenfeld, a genetic counselor and researcher in Snyder’s lab, ushered me into a small office, and then she walked me through the consent forms.
Did I understand that DNA sequencing might yield “actionable” results, such as BRCA mutations for breast and ovarian cancer, the problem that had famously spurred Angelina Jolie to undergo a preventive double mastectomy? Did I understand the test also might reveal problems I can’t do anything about, such as the APOE4 gene, which elevates the risk of Alzheimer’s? Did I want to learn all findings, only actionable ones, or none and simply donate the data to research?

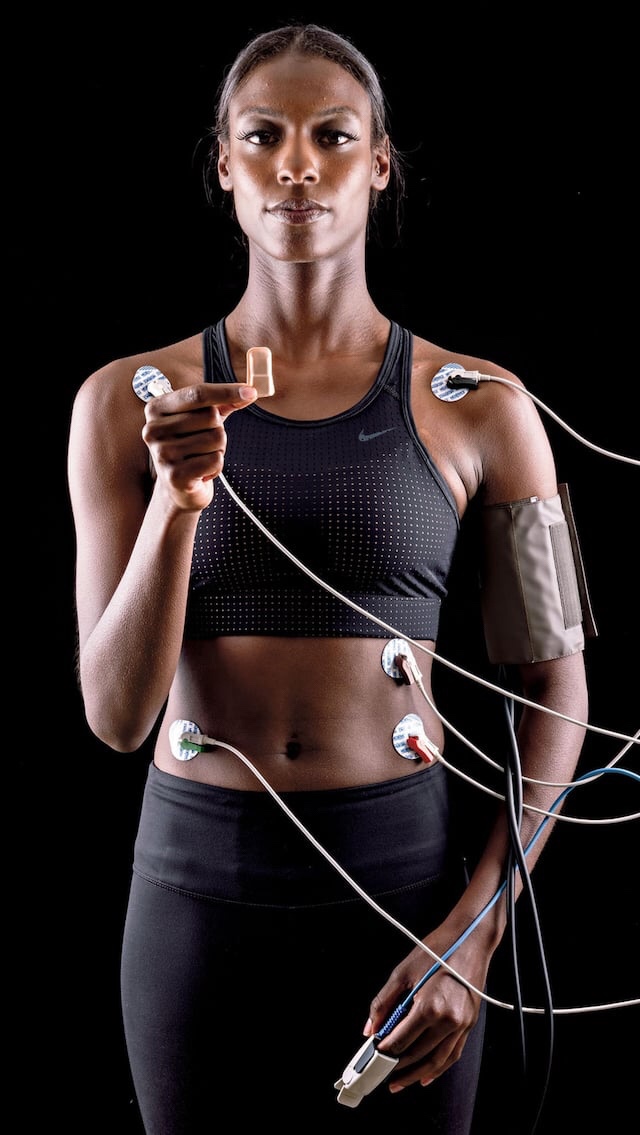
Top: Through dramatic advances in technology, miniature wireless devices are replacing bulky monitoring machines to track patient vital… Read More
SOURCE: JOHN A. ROGERS RESEARCH GROUP AT THE SIMPSON AND QUERREY CENTER FOR BIOINTEGRATED ELECTRONICS AT NORTHWESTERN UNIVERSITY (TOP)

I checked the boxes to tell me all. My stomach surprised me by knotting in protest. Nurses swabbed the inside of my nose and cheeks and drew 16 vials of blood.
At odd moments while I waited for the results, my mind replayed my father’s 10-year descent into dementia. Was that my genetic legacy? To stop brooding, I’d turn my mind to my mother. At 94, she lives on her own in New York City, gets around by bus and subway, and dances and plays mah-jongg at senior centers. The promise of genome testing is to give people more control over their health. But for the first time I came to think of mine as an inheritance—maybe a lucky one, or not, but disconcertingly beyond my command.
The DNA test found nothing bad, Dagan-Rosenfeld said right off the bat when we went over the results by video. I was grateful. But I didn’t feel as relieved as I’d expected. By then I’d learned how far science has to go before it understands what DNA can tell us.
I did learn that I metabolize some drugs poorly, including clopidogrel, so I should ask for alternatives if a doctor recommends them.

FROM DNA TO BIG DATA
In freezer trays cooled to -112°F, the UK Biobank stores blood, urine, and saliva samples from more than 500,000 people. A robot retrieves specimens that scientists use to find links between genetic variants and disease.
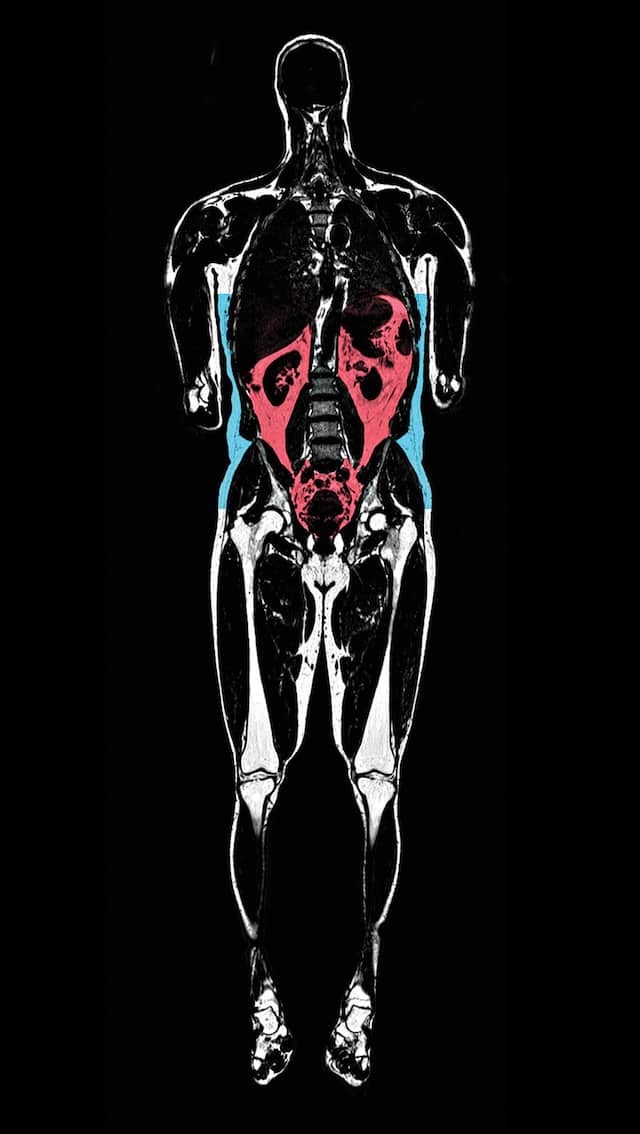
MRIs performed on some Biobank participants create 3D images that show subcutaneous fat under the skin in blue and visceral fat around the organs in red. Visceral fat has been linked to diseases. (See graphic below.)
AMRA MEDICAL AB
Snyder has turned up important genetic findings in about 17 percent of his volunteers. One had been on medication for type 2 diabetes for years—wrongly, it turns out, because he has a rare, inherited form of the illness. Another has a mutation for cardiomyopathy, a disease of the heart muscle that often goes undetected until it causes death.
For five years Snyder managed to keep his diabetes in check. Then his blood sugar crept up. Although he tried different diets and lifting weights, it continued climbing. He started taking medication. After a few months it didn’t appear to be working either. He thinks his lifestyle adjustments forestalled the problem. But his saga holds a cautionary note. Even the latest precision medical technology—and a near-religious devotion to maintaining health—may not override vulnerabilities embedded in DNA.
Genomically speaking, we’re more than 99 percent alike—but separated, on average, by millions of genetic variations. At last count, scientists had cataloged 665 million, ranging from big alterations to differences in one of the nucleotides that make up DNA.
Which variants are harmless quirks, and which pose dangers? Like parents staring at tiny toy parts and baffling assembly instructions, scientists have barely begun to figure it out.
The challenge is illustrated by an experiment conducted at Vanderbilt University. Investigators studied 2,022 people and identified 122 rare variants in two genes known to be associated with heart-rhythm abnormalities. They asked three labs to determine which variants cause the irregularities. One lab selected 16; another, 24, and the third, 17. The labs all agreed on only four. The researchers then compared the lab assessments with people’s health records and found that almost no one with potentially worrisome variants had abnormal heart rhythms.
To understand what the DNA code is saying requires huge studies over years, because risky mutations are rare and their related illnesses may take a long time to develop. The National Institutes of Health recently kicked off All of Us, part of its Precision Medicine Initiative, to collect DNA and other health information from a million people. The Dubai Health Authority plans to create a genomic database of the emirate’s three million residents.
Leading the pack on such megastudies is the UK Biobank. Tucked into an unremarkable industrial strip in Stockport, England, the biobank holds the medical secrets of 500,000 British volunteers, ages 40 to 69. A freezer, the width of a two-lane road and nearly two stories tall, stores samples of their blood, urine, and saliva—10 million specimens in tiny tubes, stacked in trays and stamped with bar codes to protect anonymity. A yellow robot trundles along a track, plucking specimens for studies. The air is compressed and dried so thoroughly to prevent frost that even standing outside the freezer, looking at it through a glass wall, my skin felt more like old leather than a living organ.
start interactive embed
YOUR GRAPHIC CODE GOES HERE SAMPLE STYLES. REMOVE ! TO SEE THIS Remove the following line if NOT using ai2html Generated by ai2html v0.78.0 – 2018-12-05 17:12 ai file: ngm-1901-precision-medicine-bmi_ai2html.ai
Artboard: Desktop-Medium Artboard: Mobile
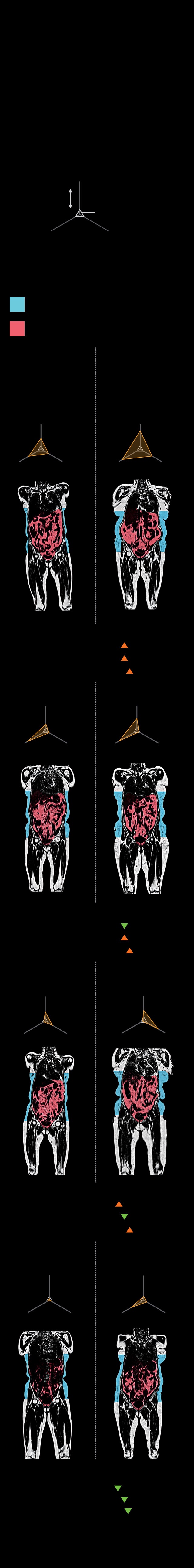
IT’S NOT THE FAT—
IT’S WHERE IT’S AT
Body mass index (BMI) has long been used to
measure obesity and health risk, yet it can’t
explain why an overweight person might be
healthy or why a seemingly healthy person
might get heart disease or diabetes. New MRI
data show that when it comes to fat, where it’s
located matters most.
FAT DISTRIBUTION
Visceral
more
Average
disease-free
individual
less
Liver
Muscular
Subcutaneous fat, under the skin, can be less
harmful than visceral fat, found around the or-
gans. Visceral, liver, and muscle fat (orange
lines, below) are linked to metabolic diseases.
Subcutaneous
Visceral
Obese
Four women with
a BMI of 32 but
different fat
distribution
Normal weight
Four women with a
BMI of 24 but differ-
ent fat distribution
Probability of:
High
Heart disease:
Type 2 diabetes:
Metabolic disease:
Probability of:
Low
Heart disease:
Type 2 diabetes:
Metabolic disease:
Probability of:
Heart disease:
Type 2 diabetes:
Metabolic disease:
Probability of:
Heart disease:
Type 2 diabetes:
Metabolic disease:
KURT MUTCHLER AND JASON TREAT, NGM STAFF;
KELSEY NOWAKOWSKI
SOURCES: UK BIOBANK; JENNIFER LINGE, AMRA MEDICAL AB
End ai2html – 2018-12-05 17:12 END GRAPHIC CODE
end interactive embed
Biobank computers link to the health records of participants, because the clues in DNA reveal themselves only when researchers can tie gene variants to traits and ailments in people. “Everybody unfortunately will be informative in the long term,” said Rory Collins, the biobank’s chief executive and principal investigator. “But only a small portion of people will be informative about a particular disease.” The biobank has genotyped tissue from every donor. The process, commonly used by consumer DNA test companies, scans the genome for specific variants. The biobank is now working with a pharmaceutical consortium to sequence every donor’s exome, the protein-coding portion of the genome. Genotyping can find oddities and defects that researchers know to hunt for; sequencing can unearth new ones.
More than 4,000 researchers around the world are using the biobank’s data trove to study the genetics of such conditions as cancer, osteoporosis, and schizophrenia and such habits as using marijuana and being a night owl.
The research, though, has limited applications to diverse populations because it tracks a largely white group. Other big genetic databases have the same drawback. A 2009 analysis of studies examining the links between genes and disease found that 96 percent of the participants were of European descent. Seven years later, University of Washington investigators reported some improvement, mostly because more studies were being done in Asia. Stanford researchers have warned that unless scientists study humanity in all its diversity, genomic advances will benefit only “a privileged few.”
Nevertheless, researchers are using the data to push the bounds of personalized medicine. Scientists at the Broad Institute in Cambridge, Massachusetts, recently unveiled a personal risk scorecard of sorts—algorithms that calculate the odds you’ll develop five serious, common ailments: heart disease, breast cancer, type 2 diabetes, inflammatory bowel disease, and atrial fibrillation.
The scorecard builds on an unsettling discovery: Many people have numerous mutations that each pose negligible risk but cumulatively present a problem. For instance, in breast cancer these little mutations collectively are as dangerous as a BRCA1 mutation and far more common, said Sekar Kathiresan, who led the research. Many people harbor these bundles of defects and don’t know it. In the not too distant future, Kathiresan said, doctors will use systems like this to score people’s risks, perhaps even at birth.

CONCEPTION’S PERFECT TIMING
After in vitro fertilization, Carolyn Bilson holds up her ultrasound marked “G.L.” for good luck by Ilan Tur-Kaspa (in blue), the medical director of the Institute for Human Reproduction in Chicago. Bilson and her partner, Tim O’Brien, conceived Westley (below) with personalized embryo transfer.
COURTESY BILSON/O’BRIEN FAMILY

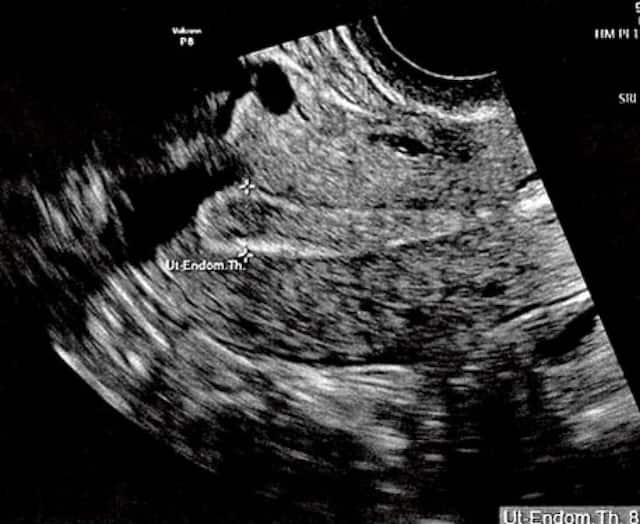

Top: The ultrasound image of Bilson’s uterus shows her endometrial lining is 8.9 millimeters thick, usually enough for implantation. But the genetic testing… Read More
COURTESY BILSON/O’BRIEN FAMILY

Westley was conceived with personalized embryo transfer. Tur-Kaspa sampled 238 genes from Bilson’s uterus to determine when she would be most receptive to implantation. An interim study found a 24 percent rise in IVF pregnancy rates with this method.
One afternoon last spring I sat down at a monitor at the Cedars-Sinai Board of Governors Regenerative Medicine Institute in Los Angeles to see a magnified view of cells under the microscope. A few months earlier, these blobs were blood cells collected from an adult donor. Scientists engineered them into primitive stem cells. And then a team led by Clive Svendsen, a neurochemist and the institute’s director, turned them into a rudimentary version of the spine—nerve tissue bearing the donor’s genetic signature. “Seems like science fiction, right?” he said. “Not long ago, it was science fiction.”
The point of this cellular shape-shifting is to create models for studying disease. Not some generalized system, but a working model of a specific patient’s specific disease. So an ovarian cancer researcher in Svendsen’s lab wants to synthesize mini-versions of fallopian tubes from the blood of an ovarian cancer patient. The gut team will make intestinal tissue from the blood or skin of children with Crohn’s disease. Svendsen studies Parkinson’s and other neurodegenerative conditions, so his team produces brain and spinal tissue.
The scientists can start with almost any adult tissue. They reprogram it, using proteins involved in gene expression to turn back the clock and convert the mature cells to embryo-like ones. The reprogrammed cells, called induced pluripotent stem cells, are then placed into a brew of growth factors and other proteins. The recipe is crafted precisely to synthesize any functioning tissue a researcher wants.
Once they create it, the scientists pull the tissue apart and lay the cells onto a chip—a translucent plate about the size of a memory stick. Made by Boston-based Emulate, the chip is lined with tiny channels that carry blood and nutrients to the cells and help them mature.
Svendsen said the model will be valuable for testing new drugs and predicting how a patient will respond to a given treatment. Figuring out which drug works best is often a miserable process, he said, citing epilepsy as an example: “We put kids through three months of hell, trying one drug after another. With the chip, you can just put a different drug on every day until you find the one that shuts down the seizure.”
Some critics believe reprogrammed cells on a chip offer only a limited view of what’s happening in the body. I asked Svendsen how he’d know, for instance, if an epilepsy drug was toxic to the liver or heart. Simple, he said. His lab would use stem cells to create mini-versions of those organs and then test them with the medication.

STUDYING PATIENT CELLS
Researcher Samuel Sances “seeds” a plastic chip with spinal cord cells from a patient with amyotrophic lateral sclerosis (ALS) research at Cedars-Sinai. This new tool, utilizing chip technology from Boston-based Emulate, allows researchers to investigate why the patient’s neurons are dying and help develop experimental treatments. “I believe that someday we’ll be conducting clinical trials inside an incubator—not in a doctor’s office,” Sances said.

By putting the chips with human cells under a microscope, Sances and his colleagues can observe neurons firing in real time (top photo below) or take pictures to analyze later (bottom photo below). Their aim is to create chips that will be able to predict how different drugs will work for an individual patient.
Play Video
Imaged through a microscope, a video of a chip with brain cells reveals hundreds of neurons… Read More
Play Video
Cells grown on a chip form a human intestinal lining with the same folds found in the organ, allowing… Read More
Just how far can cell and gene technologies push the limits of medicine? Shoukhrat Mitalipov’s laboratory at Oregon Health & Science University in Portland hints at where we might be headed. Mitalipov, a native of Kazakhstan with a boyish crop of black hair and a wrestler’s build, used the gene-editing tool Crispr-Cas9 to alter the DNA of human embryos.
Mitalipov and his international team cut a segment in the paternal gene to remove a mutation associated with the potentially fatal heart condition hypertrophic cardiomyopathy. They made the Crispr cut as they fertilized eggs from healthy donors with sperm from a man who has the disease. If these embryos could grow into babies, they wouldn’t have the disease or pass the genetic defect down the family line. Mitalipov, director of the university’s Center for Embryonic Cell and Gene Therapy, had no intention of carrying the experiment that far. The scientists grew the embryos for about three days, then removed the cells for further analysis.

Tinkering with embryo genomes and changing the gene pool of future generations was long considered taboo, but in 2015, researchers in China reported using Crispr on nonviable human embryos to modify the gene for beta-thalassemia, a potentially deadly blood disorder. Cutting the gene introduced more damage than it fixed. Mitalipov discovered no such problem. His repair technique didn’t work in every case, but he believes that with more refinement it could be used to eliminate any of the 10,000 diseases associated with single mutations.
Whether or not his method bears out, the scientific community is coming to accept the inevitability of embryo modification. A 2017 report from the National Academy of Sciences and the National Academy of Medicine concluded that a clinical trial might be permitted, though only after more research and only in dire medical cases. The technology to do it is developing fast, and perhaps the impulse to stretch the bounds of possibility is coded in our genes.
As Mitalipov led me through a small room where his team experiments on embryo genes, I asked whether he worries that the work could lead to editing embryos for traits that parents prize. He flicked his hand in exasperation. “I don’t do gene editing. I don’t do manipulation,” he said. “I do correction. Is that wrong?”

In 1978 the first “test-tube” baby, Louise Brown, also triggered anxiety about designer babies. Since then more than eight million babies have been born through in vitro fertilization and other reproductive technologies. The first heart transplant, in 1967, sparked fears that doctors would prematurely end the lives of comatose patients to harvest their organs. Now thousands of desperately ill patients around the world undergo heart transplants every year.
Even the simple home pregnancy test, available on any pharmacy shelf, set off an outcry when the FDA approved the first one in 1976. Some doctors insisted women would get too emotional about the results. A medical technologist, writing in the American Journal of Public Health, called for legislation “to limit the use of such potentially dangerous kits.”
The fears faded as these breakthroughs became commonplace. The same thing may happen as DNA sequencing, gene editing, and other once unimaginable technologies become indispensable and progress saves lives. But the precision medicine revolution is unlike any other we’ve seen. It allows us to know what has always been unknowable about our bodies and to peer into our medical future. It moves science into a new realm of biological manipulation—and repair.
Judy Perkins, who is alive today because of advances in immunotherapy and gene technologies, believes the world should be clear-eyed about the power science has unleashed. “It’s like nuclear energy,” she said. “If it gets out of control, it can be really, really ugly. And if you harness it right, it’s great.”











